
 HOME
HOME CONTACT
CONTACT SUPPORT
SUPPORT SENSORYTEST.COM
SENSORYTEST.COM |
 HOME HOME CONTACT CONTACT SUPPORT SUPPORT SENSORYTEST.COM SENSORYTEST.COM |
|
Sensory testing professional's favorite virtual desktop. Say hello to SIMS Cloud Systems. From any device, any laptop, any iPad and even your cell phone. Log in from anywhere anytime. Inquire today. 
|
Mobile devices real test example Point your cell phone camera Easy for your respondents 
Link |
 VIEW REPORTS
VIEW REPORTS |
 QUESTION CHOICES
QUESTION CHOICES |
 TEST CHOICES
TEST CHOICES |
| Data Security - Audit Trail Capability
All Panelist Data is Time/Date stamped to within 1/100th of a second. 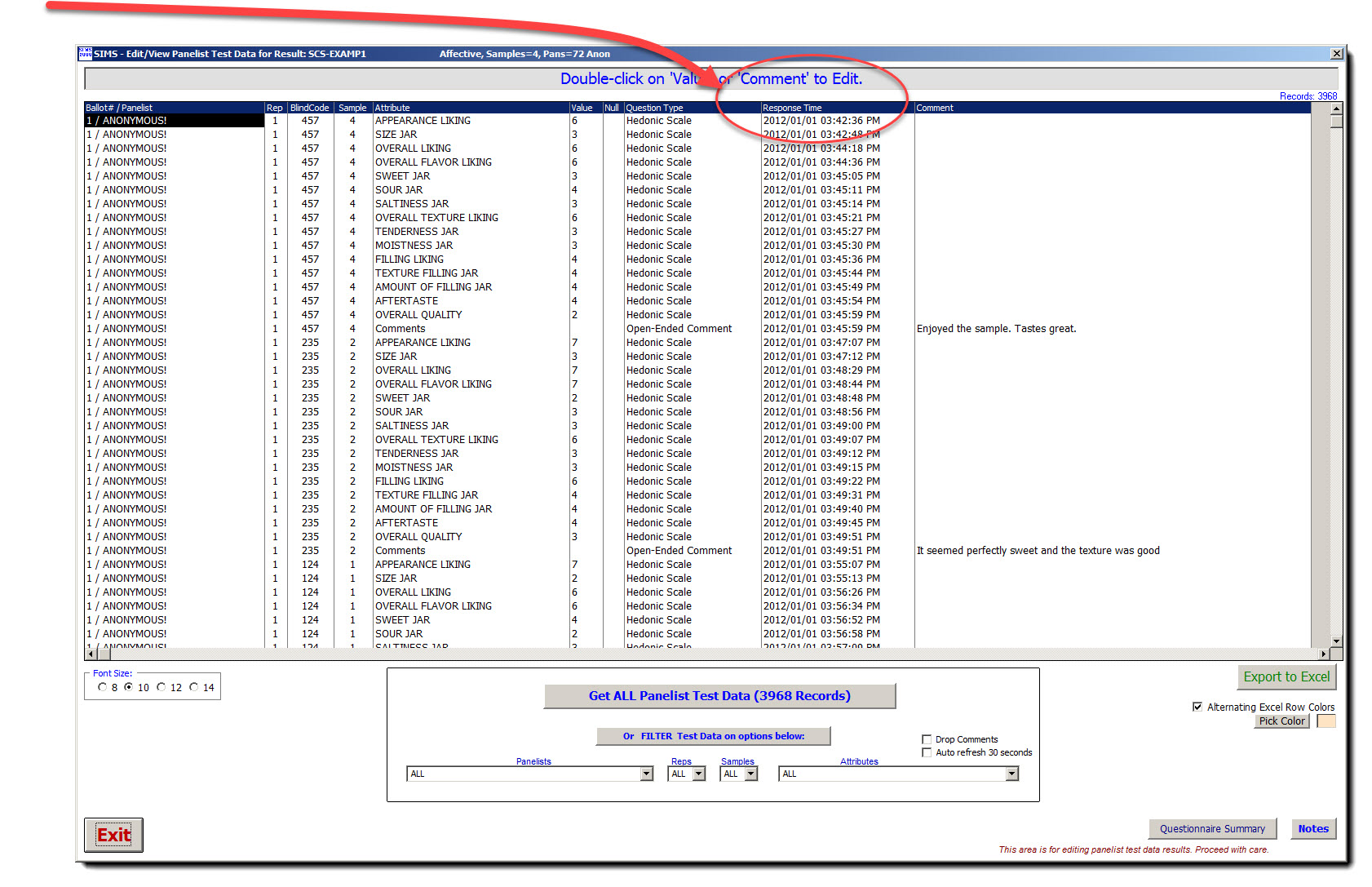
Data Security - System Event Log Reports detail a varity of events Including every SIMS Systems technican login/logout, tests started/ended, tests modified/deleted, etc. 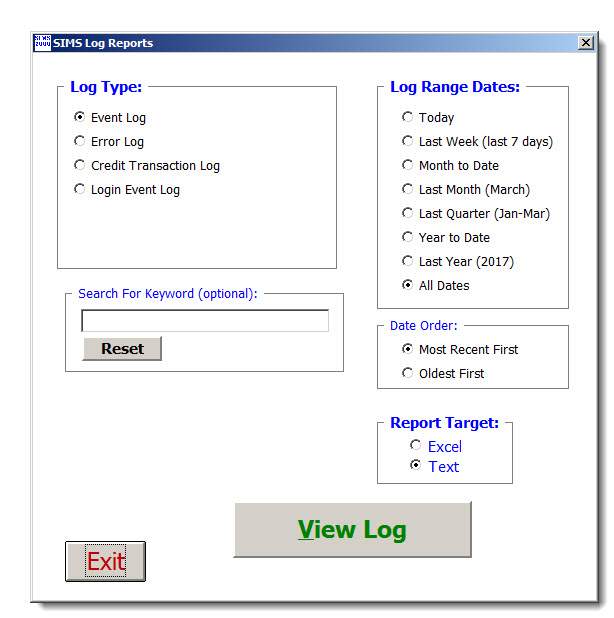
Data Security - Microsoft SQL Server SQL Server 2016/2014/2012/2008 includes significant enhancements to the security model of the database platform, providing more precise and flexible control to enable tighter security of the data. A considerable investment has been made in features that help to provide a high level of security for your enterprise data. These features include the advanced security of surface area reduction, data encryption, native encryption, authentication, granular permissions, and user and schema separations. These advancements contribute to Microsofts Trustworthy Computing initiative that defines the steps necessary to help support secure computing and helps you deploy and maintain a secure environment. Sources: Security in Microsoft SQL Server SQL Server Security Features at a Glance Data Security - FDA Good Laboratory Practices (GLP) - FDA Good Clinical Practices (GCP) SIMS Systems has always adhered to the principles of GLP & GCP data audits and data protection. SIMS Systems uses data integrity rules and validations, along with time/date stamping and detailed logs. More information: FDA Good Laboratory Practices (GLP) More information: FDA Good Clinical Practices (GCP) Data Security - CFR21 Part 11 SIMS Systems Compliance Information - CFR21 Part 11 SIMS Systems professional data security. |
